Active Pharmaceutical Ingredients
- Clopidogrel Bisulphate
Chemical structure:
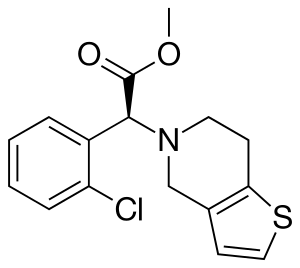
IUPAC name:
(+)-(S)-methyl 2-(2-chlorophenyl)-2-(6,7-dihydrothieno[3,2-c]pyridin-5(4H)-yl)acetate
CAS number:
113665-84-2
Therapy area:
Anti-coagulant
References:
IP/USP/Ph.Eur
Status:
Commercial sale
Mechanism of action
Clopidogrel is activated by the enzymes CYP2C19, CYP1A2, and CYP2B6 in two phases, followed by CYP2C19, CYP2C9, CYP2B6, and CYP3A. The P2Y12 subtype of the ADP receptor, which is crucial for the activation of platelets and eventual cross-linking by the protein fibrin, is thus selectively and irreversibly inhibited by the active metabolite. When a quick effect is required, a loading dosage of either 600 or 300 mg of oral clopidogrel is delivered since platelet inhibition can be seen two hours after a single dose, but the drug's
beginning of action is sluggish. - Etoricoxib
Chemical structure:
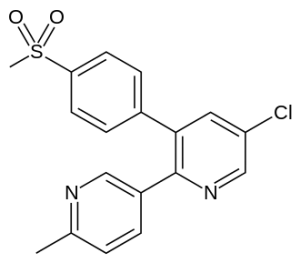
IUPAC name:
5-Chloro-6′-methyl-3-[4-(methylsulfonyl)phenyl]-2,3′-bipyridine
CAS number:
202409-33-4
Therapy area:
NSAID
References:
IH/IP
Status:
Commercial sale
Mechanism of action
Etoricoxib, like every other “coxib,” preferentially inhibits isoform 2 of the cyclooxygenase enzyme (COX-2). It selects COX-2 inhibition over COX-1 by a factor of around 106. As a result, less prostaglandins (PGs) are produced from arachidonic acid. Among the several roles played by PGs, their contribution to the chain of events leading to inflammation should be emphasised. Comparing selective COX-2 inhibitors to conventional non-steroidal anti-inflammatory medications, the latter exhibit decreased COX-1 activity (NSAID). Many sizable clinical trials with various coxibs have shown that this decreased activity is the reason for the decreased gastrointestinal adverse effects.



